 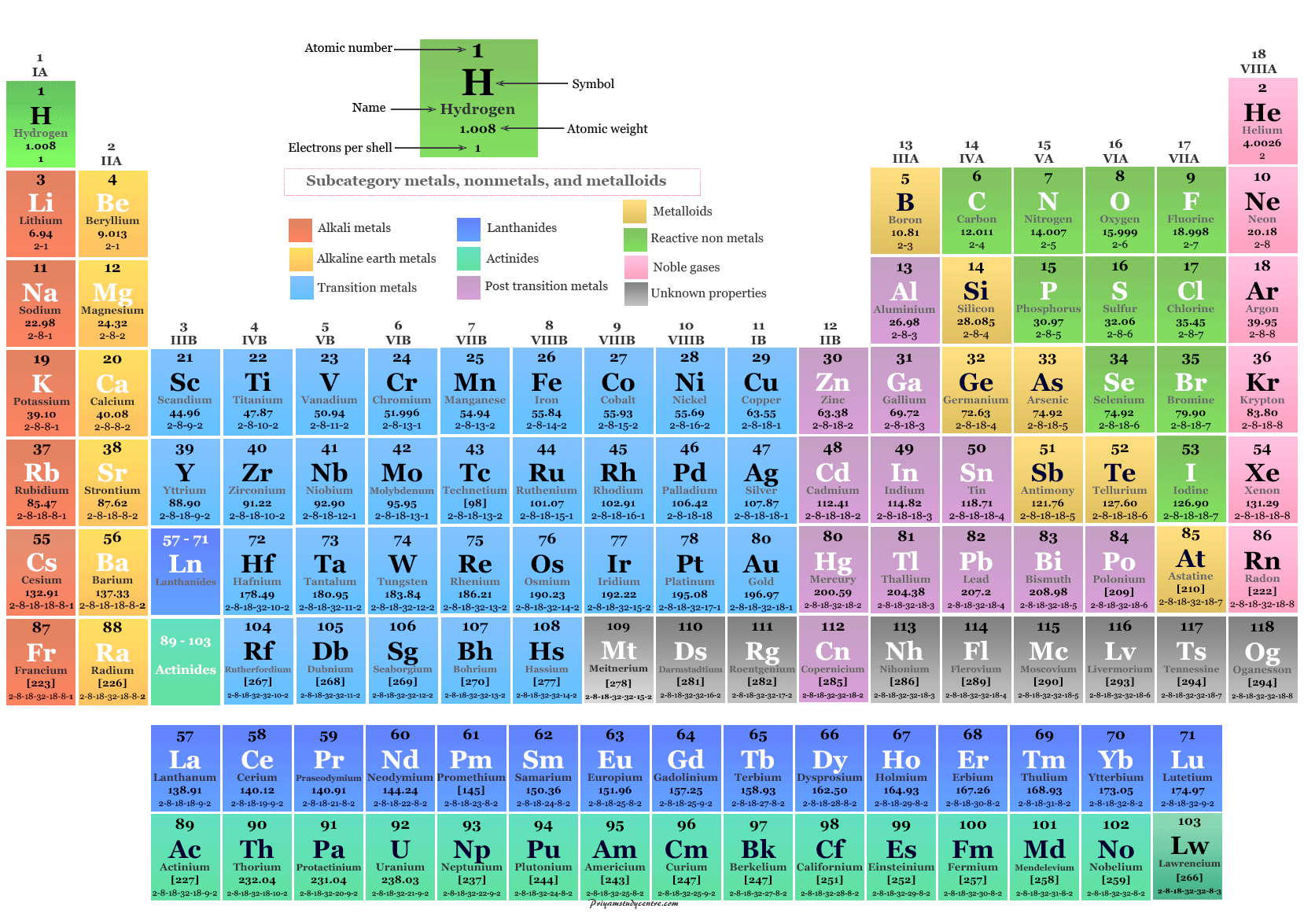
Metals are by far the largest of the three classes. They are one of three classes of elements (the other two classes are nonmetals and metalloids). 6.5: Metals Metals are elements that can conduct electricity.6.4: Modern Periodic Table- Periods and Groups.6.1: Early History of the Periodic Table.Classify each element in Conceptual Problem 1 ( Section 1.\).Other important groupings of elements in the periodic table are the main group elements, the transition metals, the lanthanides, and the actinides. Metals are lustrous, good conductors of electricity, and readily shaped (they are ductile and malleable), whereas solid nonmetals are generally brittle and poor electrical conductors. Ionization energy is denoted by the symbols IE, IP. The term is also spelled ionisation energy (British English). By definition, ionization energy is the minimum energy needed to remove the most loosely bound electron from a gaseous atom or ion. They are separated by a diagonal band of semimetals. It is a periodic table trend that increases moving across the table and decreases moving down it. Metals are located on the left of the periodic table, and nonmetals are located on the upper right. Semimetals exhibit properties intermediate between those of metals and nonmetals. The elements can be broadly divided into metals, nonmetals, and semimetals. Some of the groups have widely used common names, including the alkali metals (group 1) and the alkaline earth metals (group 2) on the far left, and the halogens (group 17) and the noble gases (group 18) on the far right. Elements that exhibit similar chemistry appear in vertical columns called groups (numbered 1–18 from left to right) the seven horizontal rows are called periods. Both the horizontal and vertical positionings of an element within the table give clues as to that element's behavior, making the periodic table a quick and useful reference for predicting how certain elements will react with each other. The periodic table is an arrangement of the elements in order of increasing atomic number. The modern periodic table organizes elements into a grid based on their atomic number. As expected, semimetals exhibit properties intermediate between metals and nonmetals. Most solid nonmetals are brittle, so they break into small pieces when hit with a hammer or pulled into a wire. Nonmetals can be gases (such as chlorine), liquids (such as bromine), or solids (such as iodine) at room temperature and pressure. Nonmetals, in contrast, are generally poor conductors of heat and electricity and are not lustrous. /periodic-table-of-the-elements-2017--illustration-769723031-5aa02f9b04d1cf00386ccf7c.jpg)
Of the metals, only mercury is a liquid at room temperature and pressure all the rest are solids. The vast majority of the known elements are metals. Metals-such as copper or gold-are good conductors of electricity and heat they can be pulled into wires because they are ductile they can be hammered or pressed into thin sheets or foils because they are malleable and most have a shiny appearance, so they are lustrous. The distinction between metals and nonmetals is one of the most fundamental in chemistry. As you might expect, elements colored in gold that lie along the diagonal line exhibit properties intermediate between metals and nonmetals they are called semimetals. Interactive periodic table with up-to-date element property data collected from authoritative sources. The heavy orange zigzag line running diagonally from the upper left to the lower right through groups 13–16 in divides the elements into metals (in blue, below and to the left of the line) and nonmetals (in bronze, above and to the right of the line). The semimetals lie along a diagonal line separating the metals and nonmetals. 
The metals are on the bottom left in the periodic table, and the nonmetals are at the top right. 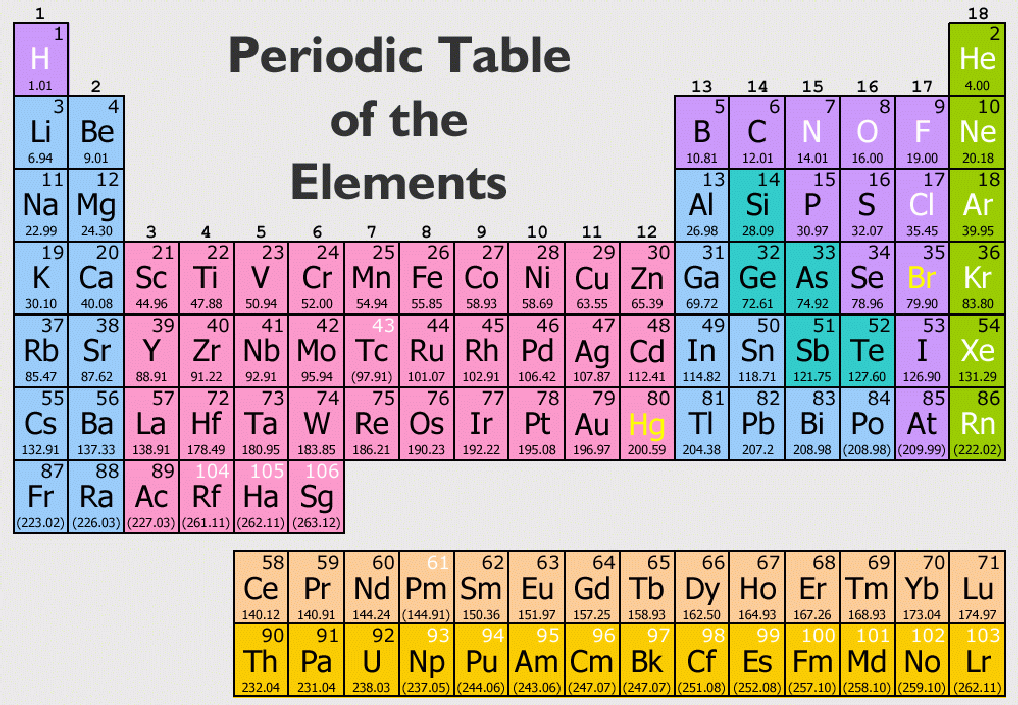
\) The Periodic Table Showing the Elements in Order of Increasing Z 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
